Ocular genetics and karma
At this year’s ARVO meeting Dr Anand Swaroop, chief of the US National Eye Institute’s Neurobiology Neurodegeneration & Repair Laboratory, received the Friedenwald Award recognising outstanding research in ophthalmology. Drew Jones found out what a career dedicated to photoreceptors has taught him about karma and discovered why people are like proteins.
How was ARVO?
At my age now, ARVO is a little bit different. It serves lot of different purposes; it's not just science, it's also social, it's networking. For students and fellows, I think it's so enriching. I can introduce them to senior people who they might be too shy or hesitant to approach… to facilitate their careers. People don't recognise how much damage Covid did when people were not able to go. Not only could they not work, but they were not able to meet others and that slowed down the careers of lot of young people by a couple of years, at least.
…And winning the Friedenwald Award and presenting the lecture?
In my opinion, it's a recognition of all the people who have contributed to my success over the years. For my lecture, rather than going through hundreds of slides of all the papers and the work that we have done over the years, I decided to present some short stories that credit my mentors who helped me understand science and, later, vision. I've not been on the bench for about 25 years now and things are changing so rapidly, so I depend on my students and fellows to educate me about new technology. I tell them that I actually work for them! I work for their success and they are the ones who will get the benefit… I just take 10% commission!
The theme of my lecture was, ‘Genes, networks and karma’. Genes and proteins don't exist in the cell alone, they are part of these large multiprotein complexes or pathways. For them to function in a normal way, everything has to be right because the substrate of one enzyme or protein is the product of another. So they're all connected and in these pathways there are really limiting steps. If any protein doesn't do its job properly, the whole network gets disrupted. It’s the same for how we work as individuals.
One message of my lecture was to follow your heart. This (he holds up a carved wooden sign) is on my desk all the time. It was given to me by the mum of a six-year-old girl who had LCA (Leber congenital amaurosis). It's in Braille as well as in English and says, ‘Thank you for fixing my eyes. If you don't, I will still be happy. But please try. Love Amy.’ It was given to me 10 years ago. I show that to every student who comes here and I tell them you need to have a reason to do what you do. I never met Amy. I don't know who she is, or where she is, but I look at those words every day and it keeps me motivated. So when people ask me when I’m retiring, I say I have promises to keep.
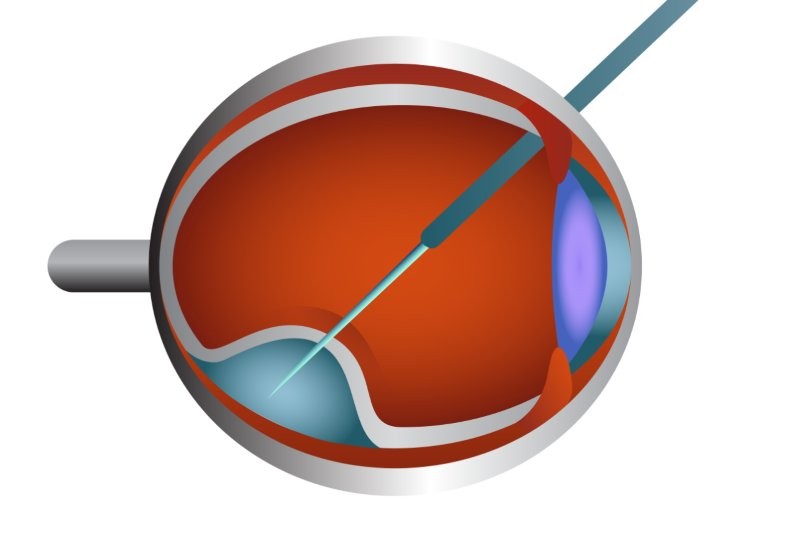
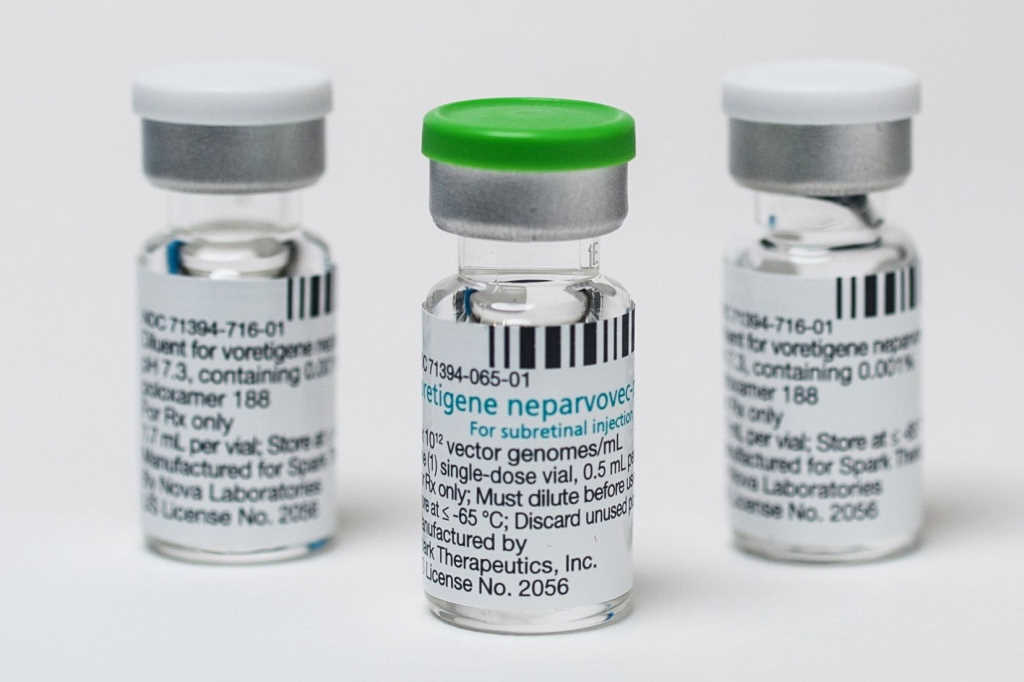
Given the cost benefits of saving sight, should governments support treatments such as Luxturna for LCA?
LCA is very complex. In general, all retinal diseases are orphan diseases, so research to find therapies and cures for them is usually funded by scientific government agencies, such as the NIH, as well as non-profit and philanthropic organisations*. While treatments like Luxturna may help a portion of the population with LCA, especially young children, there is an argument to continue researching therapies that can be used in a broader population at an affordable cost.
If you give Luxturna at a later stage, it doesn't seem to work as well in everybody and there are reasons for that. The mutation is in a gene called RPE65, which is in the retinal pigment epithelium. It's a loss-of-function mutation, which means if you put the gene back, it should function. But these tests have been done in dogs and mice – animal models that are genetically homogeneous. Humans are not genetically homogeneous. You and I have 99.9% identical genomes. But between, say, my wife and I, we have about 15 million variants. Of these, maybe 10,000 are unique to us, our parents and our kids. Now, if you have a variant in another gene of the RPE65 network, that variant might change the function of that gene by 10-20%. That’s not sufficient to cause disease by itself, but it can either decrease or increase the phenotype and affect disease progression, depending on everything else going on in that individual. I think we can design better ways to reduce the cost of drug development. My feeling is that we'd be better off doing fewer clinical trials and more well-designed biological experiments.
Does your research into AMD suggest it might one day be curable or preventable in the 15-20% of people who have a genetic predisposition?
AMD is a complex, multifactorial trait. Of its three key factors, one is genetic risk. Another big factor is age. The third, which is often ignored, is environmental factors, including lifestyle. Karma is about actions that are in your control. It's up to you to smoke or not; it's up to you to have a good diet or not. Prevention is better than cure. It's very personal to me because my father had AMD and that was more troubling for him than his cardiovascular issues. He became very depressed after he was not able to see very much, despite all the treatments.
I'm homozygous for one of the very big risk genes, but my friend Dr Emily Chew, who got the Proctor Medal at ARVO, checks me all the time and she’s a big believer in diet – she worked on the AREDS (Age-Related Eye Disease Study). So it's not 15-20% people. All of us have some risk and some more than others, but environment is a big component. So far I’m OK because I'm not a smoker, I eat a vegetarian diet and I have lot of coloured and leafy vegetables. If the disease starts when I’m 70 years old but I can slow it down by another 20-25 years, I'll probably die before the disease takes over.
Can we treat genetic susceptibility? Probably, but it's part of a network of genes, so I can’t treat that gene alone. I can try to find a small molecule drug that may impact some primary nodes within this network – a gene-agnostic therapy. My problem is that if I inject some cells into my eye, I don't know what they will do in 10 or 20 years because we don't have that much time (to study those effects). But small molecules, if you notice a problem, you can just stop. If you’re injecting the drug, it will go away through your circulation, or if it’s eye drops, you can just discontinue. I like therapies designed to work along those lines, which is what we are working on – protecting our photoreceptors before they die.
My own research focuses on understanding the metabolic pathways related to AMD, retinal degenerative diseases and glaucoma. We have a paper with a recipe for a molecule to target an important autophagy pathway we identified to protect against photoreceptor death. So my philosophy is to keep what you have rather than changing it. If you do it using a small molecule in an eye drop that costs $10-20 a month, rather than getting an injection, people can afford that. And if you're going to the Himalayas for some climbing or you’re sailing the ocean, you can take your treatment with you!
We're also trying to work out how our two-metre-long DNA is compressed within the nucleus to be able to open it up and turn on the genes in different retinal cells at a specific time and in space. We believe this will allow us to understand basic gene regulation better, so we can design a molecule to tweak that and treat more than one disease at the same time. Another very important thing is to look at how genes interact with environment. We have another paper published in Nature Communications where we talked about gene-epigenome interaction. Age and environment affect changes in the epigenome, so we are looking at how its interactions with the genome are correlated to disease.
*US$100 million of additional publicly funded costs are not evident from the $850,000 cost of a single Luxturna treatment, according to a 2019 paper in Drug Discovery Today. https://pubmed.ncbi.nlm.nih.gov/30711576/