Amniotic membrane for optometrists
In response to enquiries, New Zealand’s Optometrists and Dispensing Opticians Board (ODOB) has confirmed that using amniotic membranes (AM) for patient care is within an optometrists’ scope of practice, subject to certain restrictions.
“The board’s position is that amniotic membrane transplantation sits within the optometrist scope of practice only when application does not involve breaching the epithelium or any surgical intervention (including suturing), as this would be considered a breach of a restricted task,” it said in a statement.
The board was approached by registrants seeking clarification on the issue, said ODOB chair and Wellington-based optometrist Jayesh Chouhan. “Amniotic membrane is used in a wide range of ophthalmic conditions and I believe there is interest among optometrists to include it in their practice.”
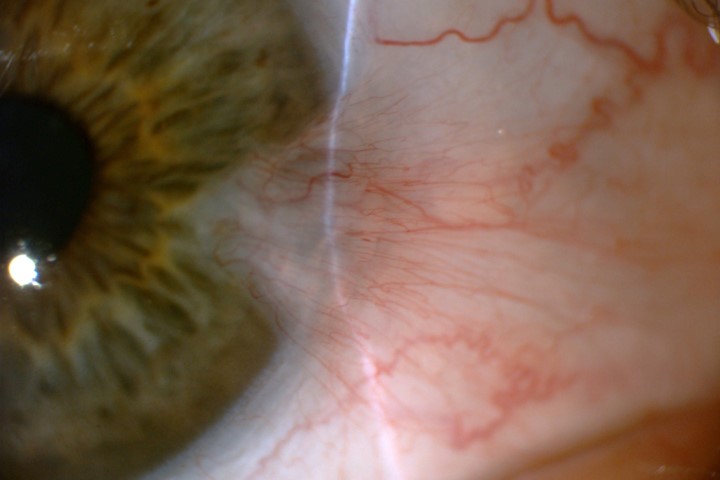
The healing properties of AM, derived from the innermost layer of the foetal placenta, are widely known and it has been prescribed as a highly effective treatment option for non-healing, chronic wounds for nearly a century, according to Dr Preeya Gupta, assistant professor of ophthalmology at the Duke University Eye Centre, North Carolina, US. More recently, AM has been embraced by eyecare professionals for its natural anti-inflammatory and anti-scarring properties. The efficacy and safety of AM for ocular surface disorders has been documented by several clinical studies, including the ‘Dry eye and amniotic membrane’ (Dream) study.
“Amniotic membrane has been shown to provide a substantial benefit in treating certain types of conjunctival and corneal disease,” wrote Dr Gupta in the American Academy of Ophthalmology’s EyeNet Magazine. “While AM has traditionally been transplanted using sutures or glue, the in-office application of sutureless AM is a less invasive alternative with promising clinical outcomes. In-office availability of AM allows clinicians to treat a variety of ocular surface conditions rapidly and effectively.”
In late 2020, Optometry Australia (OA) stated that Australian optometrists can use AM grafts as a regenerative therapy for a variety of ocular surface disease diagnoses, including dry eye, under a special access Therapeutic Goods Administration (TGA) scheme. OA’s chief clinical officer Luke Arundel said he was “pleased to see AM use become a viable option for Australian optometrists” and noted OA would be rolling out CPD in this area in 2021 through its Institute of Excellence.
Unlike New Zealand optometrists, however, Australian optometrists only have access to AM under TGA’s special access scheme, category B, which means no stock can be held in the optometry clinic. So Australian optometrists need to create an online account with the TGA and apply to use AM on a case-by-case basis, explaining each time why it is beneficial for their patient.
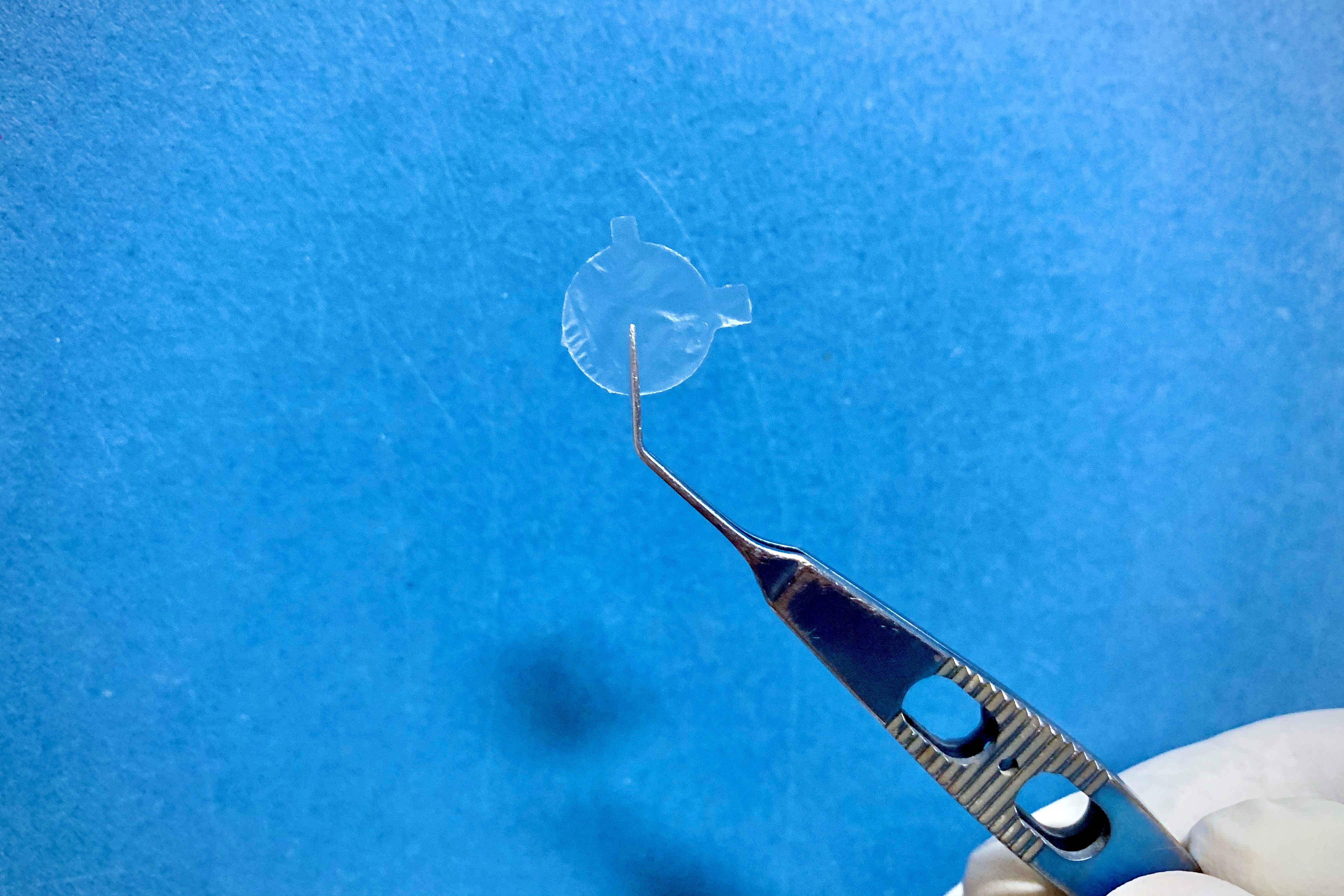
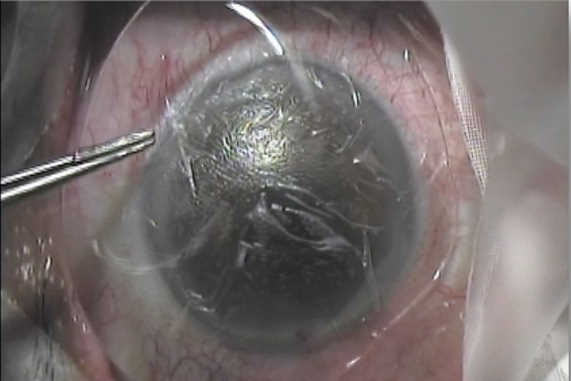
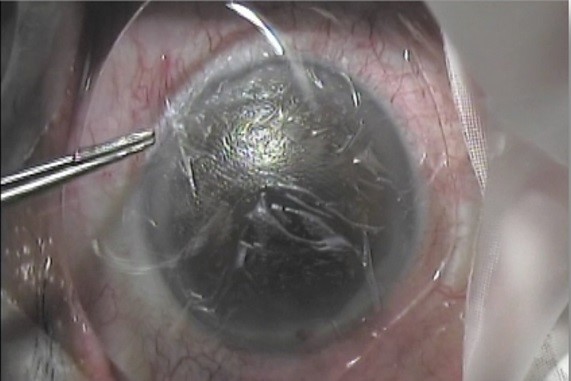
Used as regenerative therapy for the corneal epithelium, indications for AM for the optometry market include dry eye, superficial filamentary keratitis and corneal abrasions, said Jay Tardugno, founder and managing director of ISP Surgical, which manufactures and distributes AM Amniotek grafts to eyecare professionals in Australasia* and internationally. The Amniotek graft is sutureless with no adhesive and is placed on the site with a bandage contact lens over the transplant to keep it firmly in place, he told OA. “In these mild to medium cases, no corneal debridement is needed before placing the graft on the cornea, as per a bandage contact lens. These are the typical cases where optometrists would use the graft.”
For more, https://eyeonoptics.co.nz/articles/archive/amniotic-membrane-for-ded/
*Amniotek is distributed in New Zealand by Medix21 and in Australia by Designs for Vision.