Real-world effects of PBM for dry AMD
The management of non-exudative (dry) age-related macular degeneration (AMD) has historically been limited to surveillance and lifestyle modification. While the AREDS2 formulation remains the standard of care for reducing the risk of conversion to wet AMD or atrophic AMD, it offers no restorative benefit for compromised photoreceptor function or established retinal pigment epithelium (RPE) atrophy.
Recently, photobiomodulation (PBM) has garnered increasing attention as a potential therapeutic intervention for dry AMD. This non-invasive modality employs specific light wavelengths to stimulate cellular function. While initially viewed with scepticism, the evidence base supporting PBM has matured substantially recently. This shift is underscored by the recent FDA approval of the Valeda system. However, with Valeda’s New Zealand release not anticipated until later this year, the Eye-light system (available locally since July 2025) has provided an immediate avenue for clinical application and patient access.
This article briefly reviews the emerging international literature and presents findings from New Zealand’s first retrospective clinical case series using the Eye-light system, aiming to determine if reported outcomes are replicable in the real-world.
The evidence base
PBM targets cytochrome c oxidase to optimise mitochondrial function and reduce oxidative stress. In terms of specifications, the Eye-light system emits yellow (590nm) and red (630nm) light, distinguishing it from the Valeda system, which employs a tri-wavelength protocol (590nm, 660nm and 850nm). However, the total irradiance is similar between the two. Crucially, recent publications indicate that Eye-light achieves similar clinical outcomes to Valeda, while further elucidating the underlying mechanisms of action.
A recent randomised, sham-controlled trial1 evaluating Eye-light in 76 patients (152 eyes) confirmed a favourable safety profile with no significant adverse events. The study demonstrated statistically significant improvements in best corrected visual acuity (BCVA) and reductions in drusen volume and atrophy progression compared with sham. This validates the device’s capacity to induce both functional and structural change. Notably, 20% of patients achieved a ≥1-line improvement in BCVA following a single treatment cycle – a responder rate comparable to that reported for Valeda at the same interval.
Furthermore, a separate study of Eye-light shed light on the vascular mechanisms involved2. This research demonstrated that PBM induced choriocapillaris perfusion enhancement and outer retinal remodelling. The authors noted a correlation between decreased choriocapillaris flow deficits and functional visual gains, suggesting that PBM may improve the vascular supply to the compromised RPE/photoreceptor complex.
Early real-world data
To date, The Eye Clinic has treated 21 patients with the Eye-light system. We conducted a retrospective review of the 17 patients for whom minimum four-week post-treatment data was available. This real-world cohort spanned the full disease spectrum, from intermediate AMD to advanced geographic atrophy (GA). Notably, we included phenotypes typically excluded from clinical trials, such as patients with active wet AMD in the fellow eye.
The primary objective was to benchmark real-world safety and effectiveness against international data. Consistent with published data, we observed zero ocular or systemic adverse events. Functional outcomes were particularly encouraging, surpassing published efficacy endpoints: 52% of patients demonstrated a ≥1-line improvement in BCVA in both eyes.
To put these numbers into context, the following cases detail specific phenotypic responses.
Case 1: Drusenoid PED resolution with RPE preservation
Drusenoid pigment epithelial detachments (PEDs) present a significant risk of progression to photoreceptor and RPE atrophy upon collapse.
Clinical presentation: A 60-year-old woman presented with large, central drusenoid PEDs in both eyes. Natural history data indicate that the resolution of such lesions is almost invariably accompanied by atrophy of the underlying RPE and overlying photoreceptors.
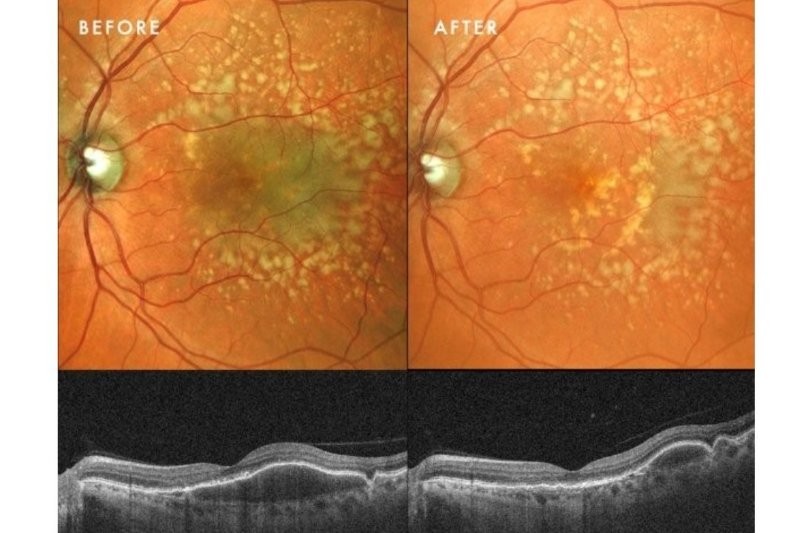
Response to therapy: Following PBM, one eye exhibited a remarkable anatomical response: the PED resolved and flattened, accompanied by a significant restoration of the ellipsoid zone on OCT. Crucially, this occurred without the expected RPE atrophy (See Fig 1 at the top of this article).
This structural success was matched functionally, with BCVA improving from 6/9 to 6/6. In the fellow eye, the PED remained structurally stable, yet she still achieved functional gain, improving from 6/30 to 6/24.
Clinical implication: This outcome suggests PBM may facilitate the metabolic clearance of sub-RPE deposits while sustaining cellular viability, potentially altering the typical course of PED collapse. Further cycles of PBM treatment and longitudinal follow-up will determine if her fellow eye eventually achieves a similar anatomical resolution.
Case 2: Prior drusenoid PED resolution with RPE atrophy
This case serves as a counterpoint to Case 1, illustrating the typical natural history of drusenoid PED collapse: progression to atrophy. It also shows the value of patient education through organistions such as Macular Degeneration New Zealand (MDNZ).
Clinical presentation: A 71-year-old man presented with a history of drusenoid PEDs first noted in 2022. Following discharge from public hospital services (reflecting the standard ‘watch and wait’ approach), he sought a referral for PBM after reviewing literature provided by MDNZ. Examination revealed that, since 2022, his PEDs had spontaneously resolved but, unlike Case 1, had progressed to moderate GA involving the foveal centre in both eyes.
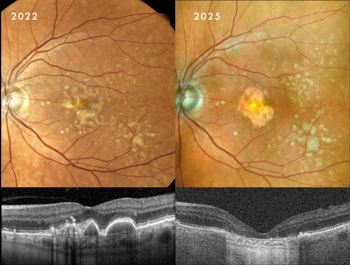
Case 2, Fig 2. This demonstrates the typical natural history of drusenoid PED resolution. There was a relatively rapid change from no atrophy initially, to moderate geographic atrophy over a 3-year period
Response to therapy: Despite the presence of established atrophy, he demonstrated a robust functional response. BCVA in the better eye improved significantly from 6/15 to 6/9, while the more severely affected fellow eye improved from 6/30 to 6/24.
Clinical implication: This aligns with the concept that PBM targets the ‘penumbra’ – the ring of stressed but viable cells surrounding atrophic areas. It demonstrates that structural loss does not preclude functional gain. By optimising the bioenergetics of the remaining photoreceptors, it is possible to stimulate visual recovery, even in eyes with foveal involvement.
Case 3: Maximising residual function in advanced disease
Patients with advanced GA are typically counselled that no medical intervention is possible, limiting management to low-vision rehabilitation.
Clinical presentation: A 76-year-old man presented with late-stage atrophy. He had previously sought multiple opinions and had even considered travelling overseas for Valeda treatment. OCT imaging revealed a precarious foveal island in the form of preserved outer nuclear layer (ONL) surrounded by dense atrophy.
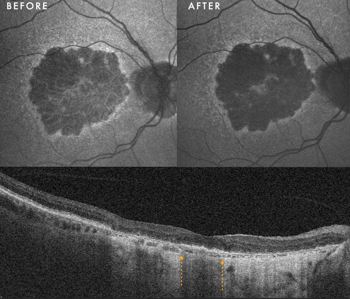
Case 3, Fig 3. Fundus autofluroescence shows a faint hyperautofluorescent signal at the fovea before PBM, which has increased significantly after PBM. The OCT demonstrates the foveal island of outer nuclear layer (between the yellow arrows) surrounded by hyper transmission/atrophy on either side. BCVA improved from 6/24 to 6/18
Response to therapy: Contrary to the poor prognosis usually associated with this phenotype, the patient achieved a 1-line improvement in BCVA (improving from 6/24 to 6/18 in the worse eye). Anatomically, we observed an increase in the hyperautofluorescent signal within the central island of residual RPE. This finding could suggest revitalisation of metabolically active tissue and potential reversal of nascent atrophic wipeout at the fovea.
Clinical implication: This case challenges the therapeutic nihilism often applied to advanced dry AMD. It suggests that as long as islands of foveal ONL persist on OCT, the tissue may remain responsive to metabolic support. PBM could potentially extend the window of useful central vision, even in the face of severe structural loss.
Patient selection
Based on this series and current literature, PBM is best considered for:
- Intermediate AMD, to potentially slow progression and reduce drusen volume.
- High-risk phenotypes, such as drusenoid PEDs, where stabilising the RPE may prevent atrophy.
- Patients with symptomatic dry AMD and early/moderate atrophy seeking to maximise residual acuity and functional vision for activities of daily living.
Conclusion
This inaugural New Zealand case series provides real-world validation of recent international data. Eye-light appears to be a safe, non-invasive therapy capable of inducing functional gain and favourable anatomical remodelling for patients with dry AMD.
While PBM is not a cure, it represents a shift from passive monitoring to active metabolic support. It appears most effective when viewed as a tool to maximise the efficiency of the remaining retinal tissue and potentially delay the structural consequences of oxidative stress.
References
1. Borrelli E, Coco G, Pellegrini M, Mura M, Ciarmatori N, Scorcia V, et al. Safety, Tolerability, and Short-Term Efficacy of Low-Level Light Therapy for Dry Age-Related Macular Degeneration. Ophthalmol Ther. 2024 Nov;13(11):2855–68.
2. Viggiano P, Boscia G, Clemente A, Laterza M, Termite AC, Pignataro MG, et al. Photobiomodulation-induced choriocapillaris perfusion enhancement and outer retinal remodelling in intermediate age-related macular degeneration: a promising therapeutic approach with short-term results. Eye. 2025 Jul;39(10):2057–63.

Dr James Leong is a consultant ophthalmologist with expertise in medical retina, uveitis and refractive cataract surgery. He is available for consultations at The Eye Clinic, Wellington and via Te Whatu Ora Health New Zealand at Wellington and Hutt Hospitals.