NLRP3 inflammasome as a key marker for DED severity
Chronic inflammation is known to play a key role in severe dry eye disease (DED), contributing to a self-perpetuating cycle of ocular surface damage. Inflammatory mediators and proteases released are key drivers of clinical signs and symptoms1,2.
Despite advances in the understanding of the disease, current anti-inflammatory therapies – including topical corticosteroids and immunomodulators, oral antibiotics, essential fatty acids and autologous serum – can offer only short-term symptom relief, with longer term use limited by side effects3,4. Research and development in this area is, therefore, ongoing in an attempt to optimise the efficacy, tolerability and safety of anti-inflammatory ocular surface therapy.
Activation of the innate immune responses in the tear film and ocular surface epithelium leads to the release of various pro- and anti-inflammatory mediators, including various interleukins and matrix metalloproteinases, with upregulated expression reported in both human and experimental DED models5–11. More recently, an emerging body of evidence supports the role of the nucleotide-binding oligomerisation-like receptor protein-3 (NLRP3) inflammasome, a key driver in the innate immune system, in the pathogenesis of DED12,13.
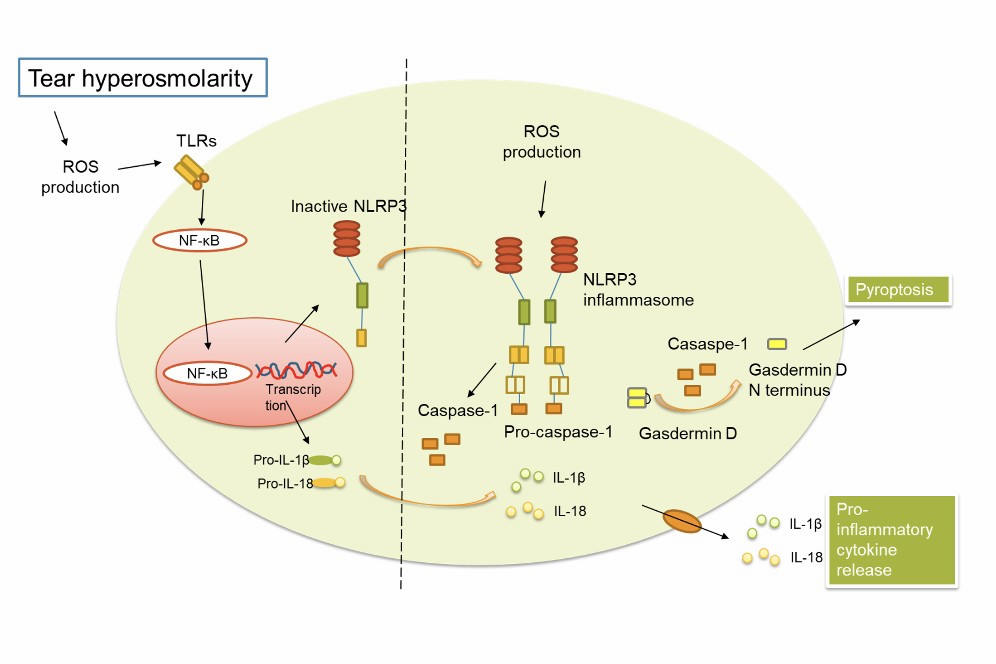
In DED, risk factors, such as ageing and desiccating environments, may trigger inflammasome activation, leading to the release of reactive oxygen species and subsequent production of pro-inflammatory mediators, such as interleukin (IL)-1β and IL-18, which regulate downstream immune response, as shown in Fig 114,15.
Recent studies have demonstrated elevated levels of inflammasome-related activities in patients with DED, particularly those with Sjögren disease13; however, evidence remains limited. To address this, my doctoral study investigated the role of NLRP3 inflammasome and its associated cytokines, as well as other inflammatory mediators, in patients with varying severity of DED.
Of the 164 participants (aged 22–84 years, 20% male) 32 were dry eye controls, 41 classified as mild, 46 as moderate and 45 as severe DED. Pooled flush tears were collected from both eyes and conjunctival epithelial cells were obtained by impression cytology from the right eyes. Tear samples were analysed using the Luminex High Performance Multiplex Assay and conjunctival samples were analysed by droplet digital polymerase chain reaction. Biomarker profiles were compared statistically across severity levels based on signs and symptoms. Tear analysis showed significantly higher levels of both downstream cytokines, IL-1β and IL-18, in the severe DED group compared to normal control, mild and moderate DED groups (p<0.05). In contrast, conjunctival impression cytology sample analysis did not show significant differences in NLRP3 inflammasome-associated gene expression across groups (p>0.05).
In summary, my study supports the critical role of the NLRP3 inflammasome in the pathogenesis of DED, particularly in severe cases. The lack of significant differences between mild and moderate groups suggests that inflammation plays a more pronounced role in advanced stages of the disease, most often associated with aqueous deficiency. Nevertheless, findings reinforce the potential of the NLRP3 inflammasome as a novel target for DED management.
References
1. Stevenson W, Chauhan SK, Dana R. Dry Eye Disease An Immune-Mediated Ocular Surface Disorder. Arch. Ophthalmol. 130, 90–100 (2012).
2. Bron AJ, et al. TFOS DEWS II pathophysiology report. Ocul. Surf. 15, 438–510 (2017).
3. Barabino S, Chen Y, Chauhan S & Dana R. Ocular surface immunity: Homeostatic mechanisms and their disruption in dry eye disease. Prog. Retin. Eye Res. 31, 271–285 (2012).
4. Kang H, et al. Cyclosporine A micellar delivery system for dry eyes. Int. J. Nanomedicine 11, 2921–2933 (2016).
5. Chen Y, et al. Interferon-gamma-secreting NK cells promote induction of dry eye disease. J. Leukoc. Biol. 89, 965–972 (2011).
6. Luo L, et al. Experimental dry eye stimulates production of inflammatory cytokines and MMP-9 and activates MAPK signaling pathways on the ocular surface. Invest. Ophthalmol. Vis. Sci. 45, 4293–4301 (2004).
7. Narayanan S, Corrales RM, Farley W, McDermott AM, Pflugfelder SC. Interleukin-1 receptor-1-deficient mice show attenuated production of ocular surface inflammatory cytokines in experimental dry eye. Cornea 27, 811–817 (2008).
8. de Paiva CS, et al. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-γ. Invest. Ophthalmol. Vis. Sci. 48, 2553–2560 (2007).
9. Lam H, et al. Tear Cytokine Profiles in Dysfunctional Tear Syndrome. Am. J. Ophthalmol. 147, 198-205.e1 (2009).
10. Massingale ML, et al. Analysis of inflammatory cytokines in the tears of dry eye patients. Cornea 28, 1023–1027 (2009).
11. Tishler M, et al. Elevated tear interleukin-6 levels in patients with Sjogren syndrome. Ophthalmology 105, 2327–2329 (1998).
12. Zheng Q, et al. Reactive oxygen species activated NLRP3 inflammasomes prime environment-induced murine dry eye. Exp. Eye Res. 125, 1–8 (2014).
13. Niu L, Zhang S, Wu J, Chen L & Wang Y. Upregulation of NLRP3 inflammasome in the tears and ocular surface of dry eye patients. PLoS ONE 10, e0126277 (2015).
14. Zhuang D, Misra SL, Mugisho OO, Rupenthal ID, Craig JP. NPRL3 inflammasome as a potential therapeutic target in dry eye disease. Int. J. Mol. Sci. 24, 1–20 (2023).
15. Dinarello, CA. Interleukin-1β, Interleukin-18, and the Interleukin-1β Converting Enzyme. Ann. N. Y. Acad. Sci. 856, 1–11 (1998).

Dian Zhuang is an optometrist who recently completed her doctoral research in the OSL, supervised by Professor Jennifer Craig and Associate Professor Stuti Misra. She now works as the Maurice and Phyllis Paykel Trust Corneal and anterior segment clinical research fellow in the Department of Ophthalmology at the University of Auckland under the supervision of Professor Charles McGhee. She also practises at a private optometry clinic, with a focus on dry eye, complex contact lens fitting, paediatric eye examinations and myopia control.