First patient dosed in Stargardt gene therapy trial
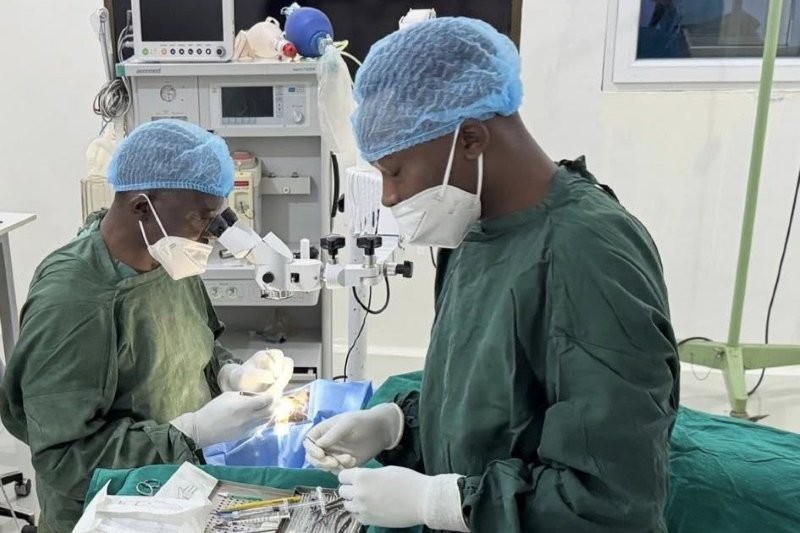
SpliceBio, a clinical-stage genetic medicine company pioneering protein splicing, has dosed the first patient in the phase 1/2 ASTRA study of SB-007, a dual adeno-associated viral (AAV) vector for the treatment of Stargardt disease.
Caused by mutations in the ABCA4 gene, Stargardt disease is the most common form of juvenile macular degeneration, leading to progressive central vision loss and ultimately blindness, with no approved treatments available. The disease has remained elusive to gene therapies due to the large size of the ABCA4 gene, said Miquel Vila-Perelló, CEO and co-founder of SpliceBio. “SB-007 is the first gene therapy in clinical development designed to restore expression of the full-length ABCA4 protein across all Stargardt disease patients, regardless of their mutations.”
Alongside the ASTRA study, SpliceBio continues to enrol patients through its POLARIS trial, a natural history study monitoring disease progression and endpoints in patients with Stargardt disease.