FDA approves macular telangiectasia therapy
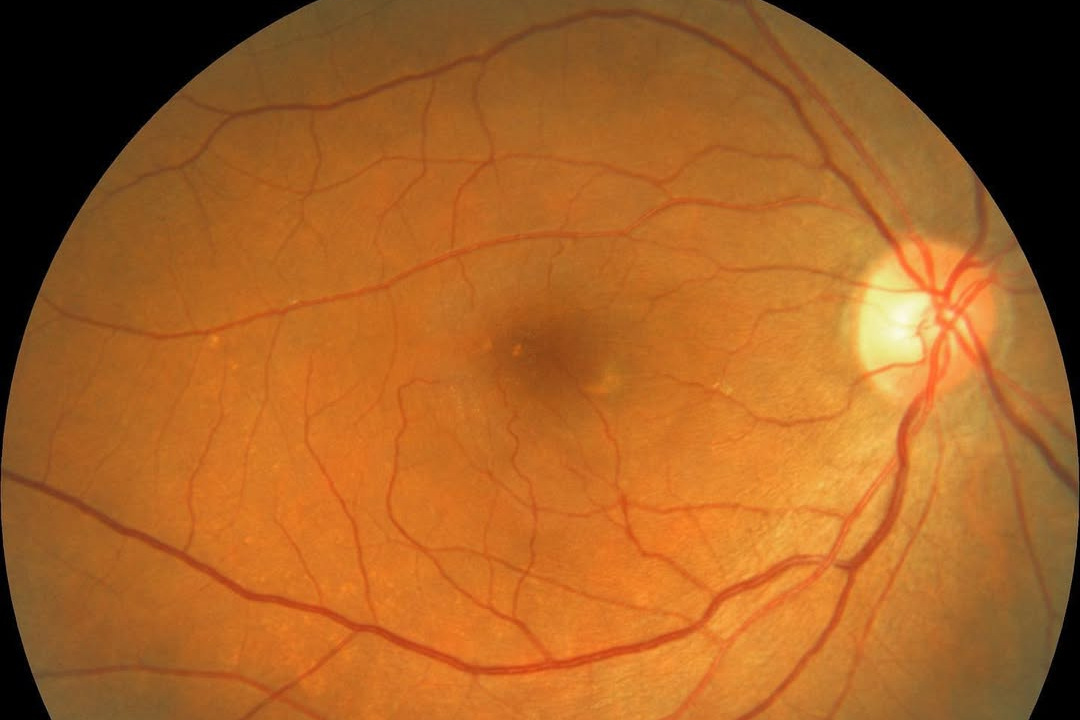
Neurotech Pharmaceuticals announced its Encelto (revakinagene taroretcel-lwey) implant has become the first US Food and Drug Administration (FDA)-approved therapy to slow macular telangiectasia (MacTel) type 2 progression.
Encelto uses encapsulated cell therapy technology to continually deliver therapeutic doses of ciliary neurotrophic factor (CNTF) to the retina. The FDA approval followed two phase 3 trials demonstrating the implant significantly slowed the loss of macular photoreceptors in MacTel patients over 24 months. “I am confident that Encelto will be able to meaningfully slow disease progression for many patients affected by MacTel, allowing them the opportunity to preserve more functional vision over time,” said clinical investigator Dr Charles Wykoff.
MacTel is often underdiagnosed due to its subtle clinical presentation, so the treatment’s approval should help raise awareness of this macula disease, said Professor Rishi Singh, from the Cleveland Clinic Lerner College of Medicine, in an interview with Ophthalmology Times. “By demonstrating the potential of sustained drug delivery to support retinal neurons, this device could (also) pave the way for future treatments targeting other neurodegenerative retinal conditions, such as geographic atrophy in age-related macular degeneration or inherited retinal diseases.”
Encelto is expected to be available to US patients from June 2025.