BTX-A as a novel therapeutic approach for DED
Traditional dry eye disease (DED) management strategies encompass artificial tears, anti-inflammatory agents and punctal plugs. However, a subset of patients remains refractory to these treatments, necessitating alternative therapeutic approaches. While dry eye can be an inadvertent side-effect of botulinum toxin A (BTX-A) applied for cosmetic purposes1, there is growing recognition of its potential to be used as a treatment to benefit patients with DED. A recent systematic review, The effect of botulinum toxin A on dry eye disease and syndromes by Amy Yen-Chi Chen, Dr Brian Sloan and myself, explored the potential therapeutic role of BTX-A in managing dry eye disease and related syndromes2. Major findings of this systematic review are presented here.
Mechanism of action
BTX-A is a neurotoxin produced by Clostridium botulinum. It functions by inhibiting acetylcholine release at neuromuscular junctions, leading to temporary muscle paralysis. In the context of DED, BTX-A's mechanism may be symptom alleviation by reducing eyelid muscle overactivity, thereby decreasing tear evaporation and improving tear film stability. Additionally, BTX-A may modulate neurogenic inflammation, contributing to symptom relief.
Clinical evidence
The review synthesises findings from 18 studies assessing BTX-A 's efficacy in DED management:
- Lacrimal gland injection: some studies have explored BTX-A injections into the lacrimal gland to reduce reflex tearing in conditions like epiphora. While this approach addresses excessive tearing, its applicability to DED, characterised by insufficient tear production, is limited.
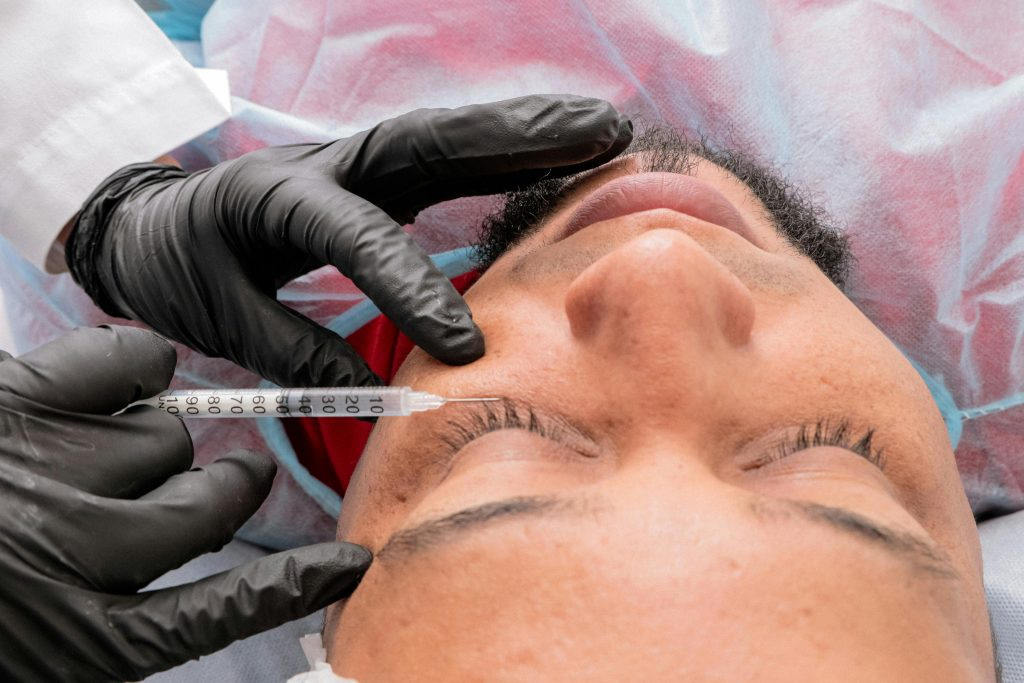
- Periocular injection: injecting BTX-A into periocular muscles, particularly the orbicularis oculi, has shown promise in reducing tear evaporation by decreasing blink-related tear film disruption. Clinical trials have reported improvements in subjective symptoms and objective measures, such as tear breakup time and ocular surface staining.
- Meibomian gland dysfunction (MGD): BTX-A's role in MGD, a common contributor to evaporative DED, has been investigated. By modulating muscle activity around the meibomian glands, BTX-A may enhance lipid secretion, improving tear film stability.
Safety and side effects
BTX-A is generally well-tolerated, with adverse effects being rare and typically mild. Potential side effects include ptosis, diplopia and transient lagophthalmos, often resolving without intervention. Proper injection technique and dosing are crucial to minimising risks.
Implications for practice
For clinicians managing DED, BTX-A offers an alternative avenue, especially for patients with significant evaporative components or coexisting MGD. For patients, it offers another possible option for those who have tried numerous therapies, such as artificial tears, warm compresses, omega-3 supplements and prescription anti-inflammatories, with little or inconsistent relief. As always in dry eye, individualised treatment plans that consider patient-specific factors and potential risks are essential for maximising therapeutic outcomes.
Conclusion
BTX-A presents a promising adjunctive treatment for DED, particularly in cases which are unresponsive to conventional therapies. Its multifaceted mechanisms – reducing tear evaporation, modulating inflammation, and improving meibomian gland function –contribute to its therapeutic potential. However, further large-scale, randomised controlled trials are necessary to establish standardised protocols, optimal dosing and long-term safety profiles.
References
1. Sullivan DA, da Costa AX, Del Duca E, et al. TFOS Lifestyle: Impact of cosmetics on the ocular surface. The ocular surface 2023;29:77-130.
2. Chen AY-C, Sloan B, Misra SL. The effect of botulinum toxin A on dry eye disease and syndromes. Clinical and Experimental Optometry 2025;1-8.

Associate Professor Stuti Misra is based at the University of Auckland. Her research interests include ocular surface diseases, anterior segment imaging, corneal innervation and diabetic retinopathy. She is a recipient of awards including the Fulbright New Zealand Scholar Award, Humboldt Fellowship for senior researchers and the Asia-Pacific Academy of Ophthalmology Achievement Award.