Presbyopia drops and SLT vs drops for glaucoma
Safety and efficacy of AGN-190584 in individuals with presbyopia: The Gemini 1 phase 3 randomised clinical trial
Dr George Waring IV et al
JAMA Ophthalmology 2022; 140:363-71.
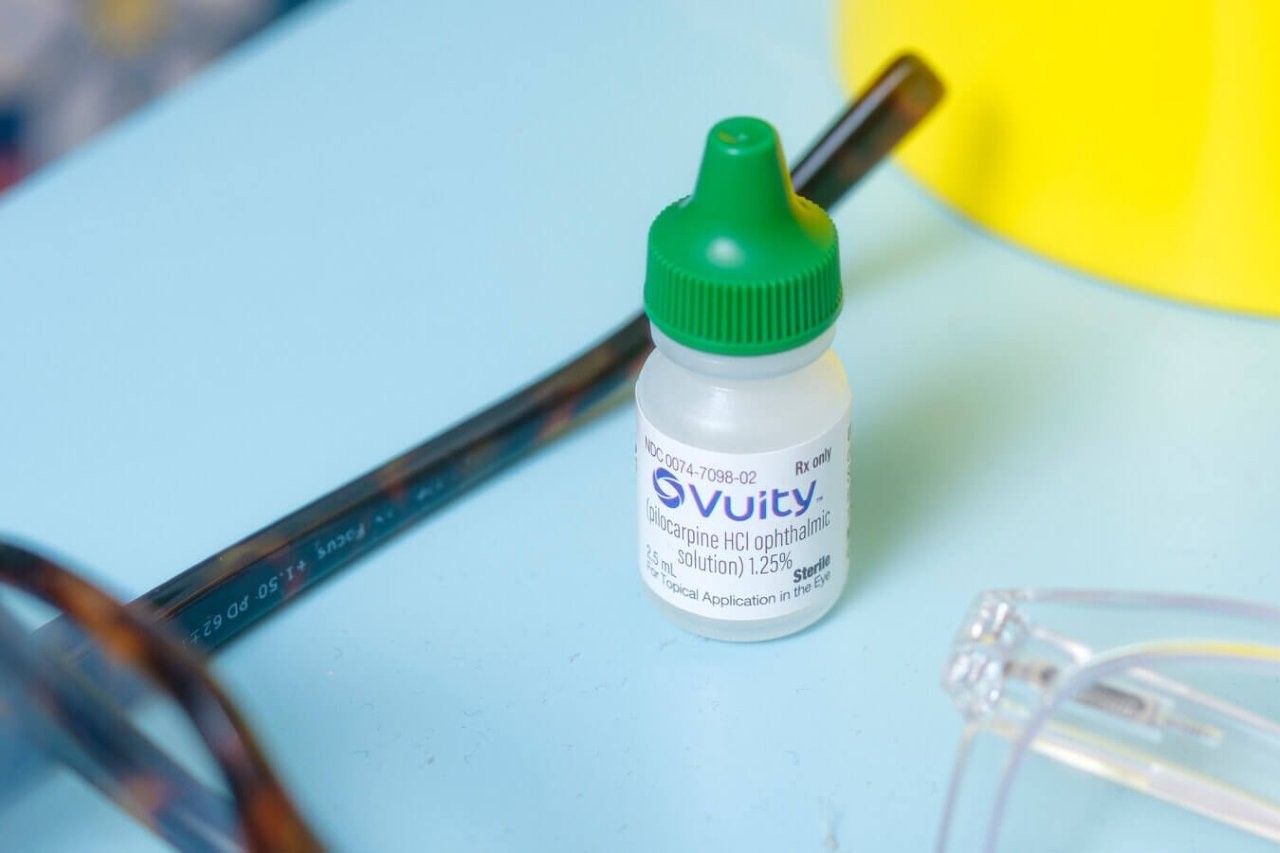
Review: This phase 3 study assessed a topical eye drop designed to improve near visual acuity in presbyopes aged 40 to 55. AGN-190584 (Allergan) is an optimised topical formulation of pilocarpine hydrochloride (1.25%) enhanced with a proprietary vehicle. AGN-190584 or the AGN-190584 formulation vehicle was administered bilaterally, once daily for 30 days.
The main outcome measures were the proportion of participants with three or more lines improvement in mesopic, high-contrast, binocular distance-corrected near visual acuity (DCNVA) at hours three and six on day 30. On day 30, hour three, the percentage of participants with improvement of three or more lines in mesopic DCNVA was 30.7% (50 of 163) in the AGN-190584 group and 8.1% (13 of 160) in the vehicle group (p< 0.001). At hour six, those percentages were 18.4% (30 of 163) and 8.8% (14 of 160), respectively (p=0.01).
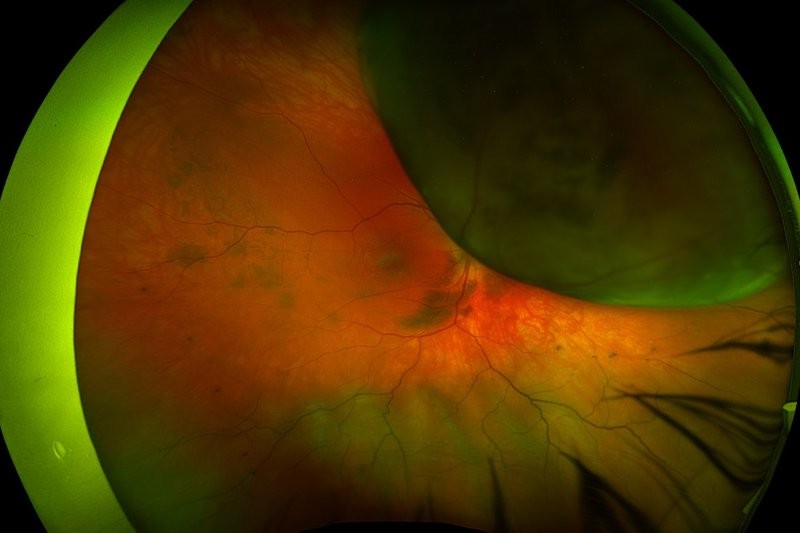
Comment: The next ‘holy grail’ of eyecare is a solution to presbyopia, particularly now that we live in a device-centric world. An eye drop that temporarily improves near vision seems a great solution. The medication in this study was pilocarpine 1.25%, enhanced with a proprietary vehicle. It is now on the market and available on prescription in the US and has received a lot of media attention there. However, pilocarpine has been used for many decades to treat glaucoma and its ability to improve near vision through miosis, resulting in improved depth of focus, is well known. This study demonstrated that the medication can statistically improve near vision for up to six hours compared to the vehicle, although the effect was relatively mild, with only 30% achieving an improvement in three lines of near acuity. Pilocarpine is rarely used these days for glaucoma due to the need for dosing four times per day and potential side effects such as headache, blurred distance vision and risk of retinal detachment.
While this study reported that the medication was well tolerated, these are all potential side effects that eyecare providers should be aware of. In particular, a recent case series reported three retinal detachments (one bilateral) after initiation of this medication¹. While this medication will likely be useful for some patients, we do not seem to have the holy grail just yet. There are several other drops currently in the development pipeline², and it will be interesting to see how these perform as they become available.
- American Journal of Ophthalmology, https://doi.org/10.1016/j.ajo.2022.05.011
- https://www.nzoptics.co.nz/articles/archive/anti-presbyopia-drops-fact-or-fiction/
SLT versus medication for open-angle glaucoma: systematic review and meta-analysis of randomised clinical trials
Chi SC, et al.
Br J Ophthalmol. 2020 Nov;104(11):1500-1507.
Review: This analysis of randomised controlled trials conducted before August 2019 compared the efficacy of selective laser trabeculoplasty (SLT)-related and medication-only treatments for open-angle glaucoma (OAG). Overall, 1,229 patients in eight trials were included.
The results revealed no significant differences between SLT-related and medication-only treatments regarding intraocular pressure (IOP) reduction and the success rate of IOP control. The SLT-related therapy group required significantly fewer medications compared with the medication-only group. The conclusions were that SLT is safe and has a lower incidence of ocular side effects compared to medications, and can be a first-line therapy for OAG.
Comment: The decision regarding when to start treatment for early or possible glaucoma can be difficult, as the clinician is often committing the patient to treatment for life. All currently available treatments for OAG involve the reduction of IOP. Traditionally, topical medication has been the first-line treatment for glaucoma, with prostaglandin analogues usually the first choice. However, the advent of SLT has presented another option that now, according to studies, is of similar efficacy to medication.
While cost and significant adverse events were not evaluated in this study, it is well known that over time, eye drops can have significant side effects including ocular irritation and prostaglandin-associated orbitopathy. In addition, studies have shown that up to half of patients on topical medications do not use them as regularly as instructed. Thus, SLT can be a favourable alternative to medications for some individuals. It is important to note, however, that some individuals do not have an appreciable IOP-lowering response to SLT and will still need further IOP-lowering medication. In addition, patients need to be counselled regarding the temporary effects of SLT and potential need to repeat treatment after one to two years.

Dr Jay Meyer is an Auckland-based consultant ophthalmologist specialising in cornea/external diseases and glaucoma. He is also a senior lecturer at Auckland University and works in private practice at Eye Institute.