Adverse effects of cancer therapy on the ocular surface and anterior segment
With a large (and growing) number of New Zealanders undergoing cancer therapy, eyecare professionals should be cognisant of the ocular complications that can result from many of these treatments to ensure their timely and appropriate diagnosis and management. Although chemotherapy remains a mainstay of the medical management of cancer, the repertoire and use of newer targeted therapies also continues to expand. With more tumour-specific effects on cell signalling and growth, this article focuses on these targeted therapies and their effects on the ocular surface and anterior segment.
Anterior uveitis
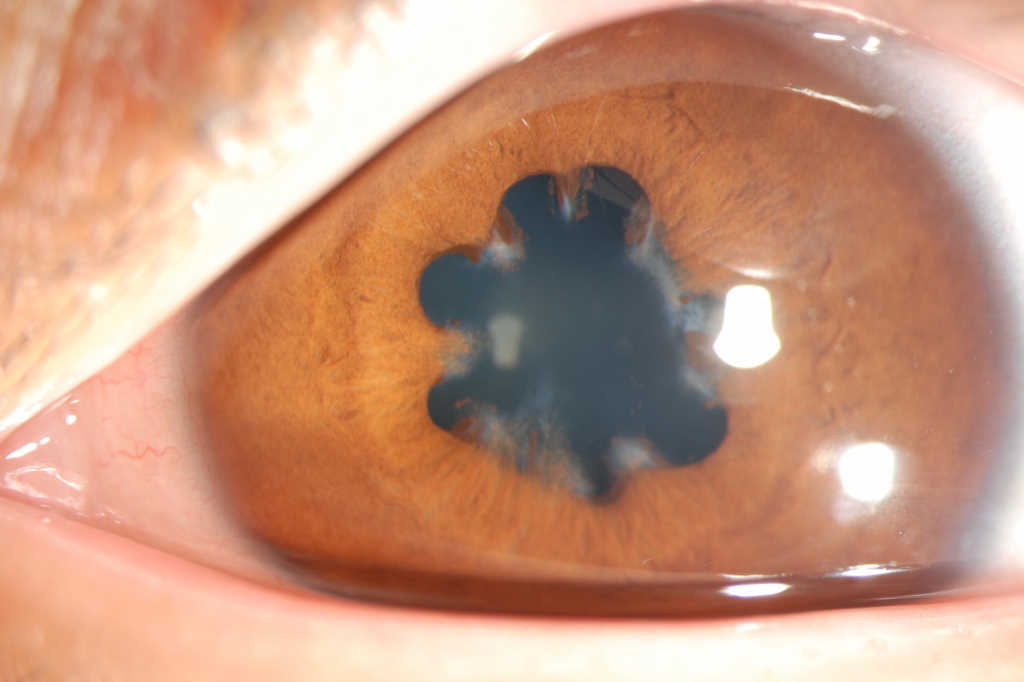
Intraocular inflammation of the iris and/or anterior part of the ciliary body, if uncontrolled, can become a serious complication of anti-cancer therapy. With circumlimbal injection and anterior chamber cells and flare commonly seen upon slit lamp examination, anterior uveitis usually causes ocular pain, photophobia and decreased vision. Drug-induced anterior uveitis has been documented for many anti-cancer agents, such as BRAF inhibitors¹. These target the BRAF enzyme, which is involved in cell growth, and includes drugs like vemurafenib, used to treat advanced melanoma in patients with a specific BRAF gene mutation. The additional immuno-stimulatory effects of BRAF inhibitors (such as increasing immune cells and releasing immunosuppression) are beneficial within the tumour microenvironment but could underpin the manifestation of uveitis in the eye.
Immune checkpoint inhibitors, such as nivolumab and ipilimumab, can also cause anterior uveitis2,3. Both of these monoclonal antibodies are indicated for the treatment of advanced melanoma and lung cancer, and bind immune cells to prevent their inactivation. Nivolumab and ipilimumab target programmed cell death protein 1 (PD-1) and cytotoxic T-lymphocyte antigen 4 (CTLA4), respectively, however, the eye may also use both molecules to maintain immune privilege. Thus, it is possible that disruption of this by immune checkpoint inhibitors could result in uveitis.
The management of anterior uveitis typically involves topical corticosteroid and mydriatic/cycloplegic agents initially, although patients should be closely monitored for worsening pain and vision loss, significantly raised intraocular pressure and progression to posterior uveitis.
Dry eye disease
Dye eye disease (DED) is one of the most common ocular sequelae secondary to the administration of anti-cancer agents, resulting from loss of tear-film homeostasis and ocular surface inflammation and damage. Often presenting with transient blurred vision and ocular discomfort, tear volume and stability investigations can also help diagnose DED. Around 35-59% of patients on aromatase inhibitors develop DED, with symptom severity increasing with treatment duration4,5. This class of therapeutics includes anastrozole, exemestane and letrozole, which may cause DED by impairing meibomian gland secretion.
Used to treat breast cancer and to prevent its development in high-risk individuals, these drugs inhibit oestrogen production, thus interfering with breast tissue growth. How aromatase inhibitors cause DED is unknown, as oestrogen’s effect on tear film physiology is disputed. Up to 31% of patients on trastuzumab (Herceptin) for breast cancer also exhibit drug-associated DED6. This monoclonal antibody is used to treat tumours overexpressing human epidermal growth factor receptor 2 (HER2), which is involved in cell growth. Trastuzumab binds and inhibits HER2, but with HER2 also expressed on the ocular surface, it could be hypothesised that trastuzumab contributes to the ocular surface damage component of DED by disrupting epithelial cell growth.
Immune checkpoint inhibitors, such as pembrolizumab (Keytruda), can also induce drug-related DED7. This monoclonal antibody is often used to treat advanced melanoma by reducing immunosuppression and supporting an anti-neoplastic immune response. However, by releasing immunoregulation in the normally immune-privileged eye, this has been postulated to contribute to the inflammatory component of DED by allowing autoimmunity and immune cell activity. In line with the management of DED due to other causes, this usually involves topical lubrication and addressing underlying abnormalities, such as meibomian gland dysfunction, using warm compresses.

Inferior punctate epithelial erosions from DED. Credit: Prof Jennifer Craig, UoA
Corneal pathologies
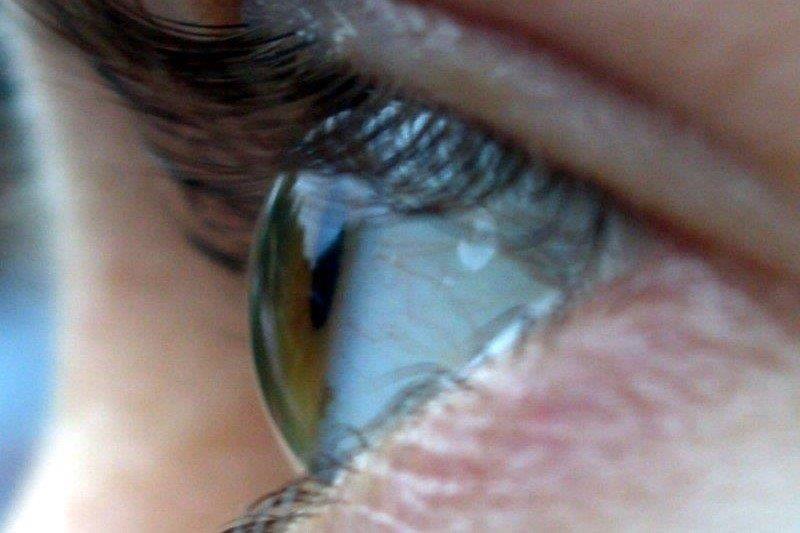
Although less common, keratitis, keratopathy and corneal ulceration and perforation have also been documented following cancer treatment. Potentially sight-threatening, some of these abnormalities can require anti-cancer drug discontinuation and are particularly associated with epidermal growth factor (EGF) receptor inhibitors.
Although these therapies aim to reduce neoplastic cell growth, many cell types on the ocular surface and in the anterior segment may also be affected. For instance, keratitis is a known side effect of osimertinib, afatinib, erlotinib and gefitinib, which are all indicated for advanced lung cancer with specific EGF receptor mutations8-11. Medsafe recommends cessation of afatinib and gefitinib therapy upon a confirmed diagnosis of ulcerative keratitis9,11. Corneal erosions related to gefitinib use have also been described and these may be dose-related12.
The interruption of EGF-dependent cellular processes in the eye by EGF-receptor inhibitors could account for some of the pathologies observed. Impaired corneal epithelial cell proliferation, adhesion and migration could affect wound healing and inflammation, potentially leading to keratitis and corneal erosions. These pathologies could also be exacerbated by diminished meibomian gland epithelial cell proliferation, through potentially inadequate lubrication of the corneal surface. Finally, impaired keratocyte proliferation, differentiation, migration and production of stromal components could affect wound healing and cause stromal thinning, and thus corneal ulceration and perforation.
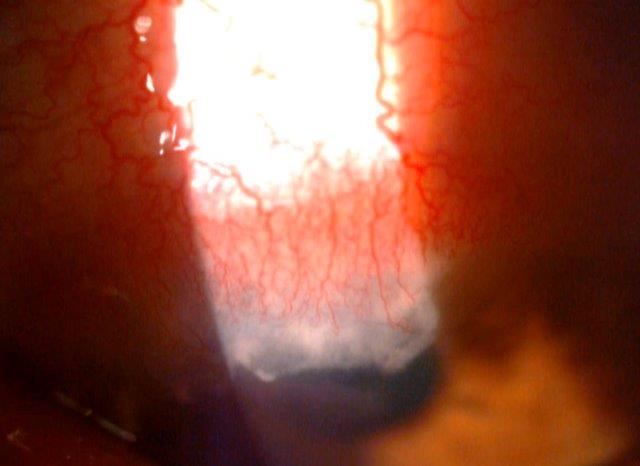
Peripheral keratitis. Credit: Dr Mohammed Ziaei, UoA
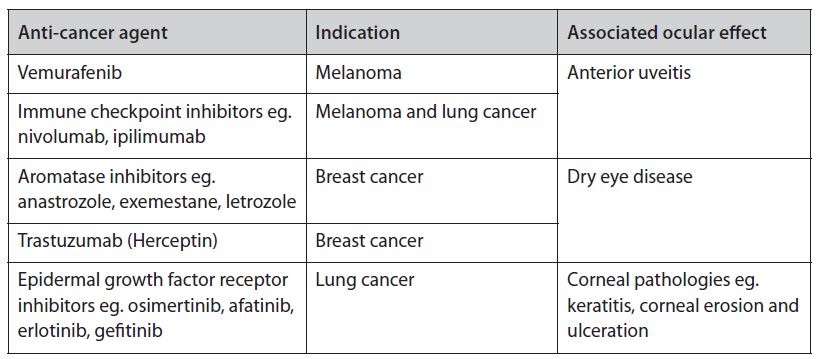
Table 1. Examples of targeted anti-cancer agents and their associated ocular effects
Other cancer treatment ocular concerns
Although outside the scope of this article, other common cancer-related treatments and associated ocular toxicities should be noted. For instance, cisplatin (one of the most widely used chemotherapeutic agents), oxaliplatin (another platinum-based compound, used in gastrointestinal cancers) and tamoxifen (used in hormonal therapy for breast cancer) can all cause a variety of visual disturbances. These treatments can also lead to posterior segment abnormalities, such as optic neuritis and retinopathy.
Corticosteroids are also often included in cancer treatment regimens to alleviate a variety of symptoms; however, their use may be associated with the development of cataract and glaucoma.
In summary, it is imperative that eyecare professionals consider anti-cancer regimens as a possible aetiology where relevant, to ensure the prompt and accurate diagnosis and treatment of drug-related ocular pathologies.
References
- Mettler C et al. Ocular safety profile of BRAF and MEK inhibitors: data from the World Health Organization Pharmacovigilance Database. Ophthalmology. 2021 May 14:S0161-6420(21)00366-3.
- Dow ER et al. Immune checkpoint inhibitor-associated uveitis: review of treatments and outcomes. Ocul Immunol Inflamm. 2021 Jan 2;29(1):203-211.
- Venkat AG et al. Local therapy for cancer therapy-associated uveitis: a case series and review of the literature. Br J Ophthalmol. 2020 May;104(5):703-711.
- Ma J et al. Prevalence of symptomatic dry eye in breast cancer patients undergoing systemic adjuvant treatment: A cross-sectional study. Breast. 2020 Oct;53:164-171.
- Inglis H et al. Dry eyes and AIs: If you don't ask you won't find out. Breast. 2015 Dec;24(6):694-8.
- Banerji U et al. Trastuzumab duocarmazine in locally advanced and metastatic solid tumours and HER2-expressing breast cancer: a phase 1 dose-escalation and dose-expansion study. Lancet Oncol. 2019 Aug;20(8):1124-1135.
- Herbst RS et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–50.
- AstraZeneca Limited. Tagrisso 40 and 80 mg film coated tablets data sheet 24 March 2021. https://www.medsafe.govt.nz/profs/Datasheet/t/tagrissotab.pdf (accessed 1 August 2021).
- Boehringer Ingelheim (New Zealand) Limited. Giotrif 20, 30, 40 and 50 mg film coated tablets data sheet 18 January 2021. https://www.medsafe.govt.nz/profs/Datasheet/g/giotriftab.pdf (accessed 1 August 2021).
- Roche Products (New Zealand) Limited. Tarceva 25 mg, 100 mg, and 150 mg film coated tablets data sheet 12 February 2018. https://www.medsafe.govt.nz/profs/Datasheet/t/tarcevatab.pdf (accessed 1 August 2021).
- AstraZeneca Limited. Iressa 250 mg film coated tablets data sheet 6 October 2020. https://www.medsafe.govt.nz/profs/Datasheet/I/Iressatab.pdf (accessed 1 August 2021).
- Tullo AB et al. Ocular findings in patients with solid tumours treated with the epidermal growth factor receptor tyrosine kinase inhibitor gefitinib ('Iressa', ZD1839) in Phase I and II clinical trials. Eye (Lond). 2005 Jul;19(7):729-38.

Dr Isabella Cheung is a research fellow in the Department of Ophthalmology at the University of Auckland. Her current research interests include pharmacoepidemiology and health services in ophthalmology, using national government data sets.