The case for funding Luxturna in NZ
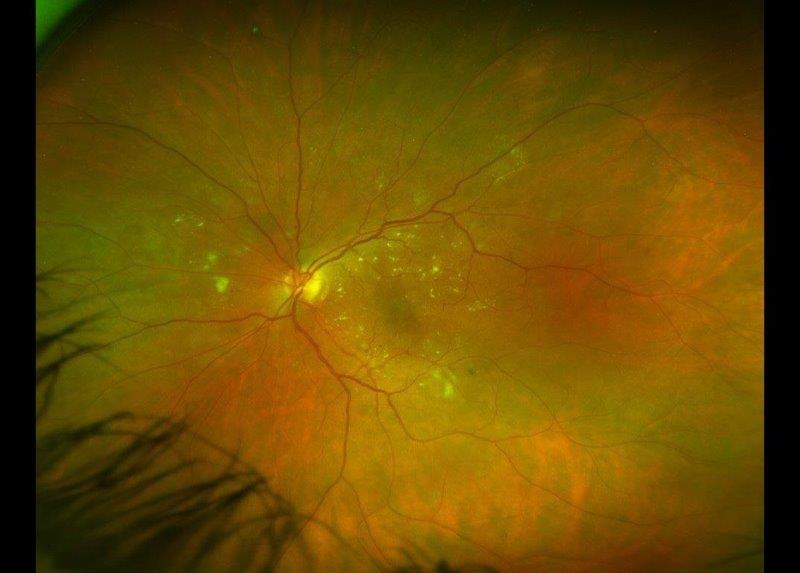
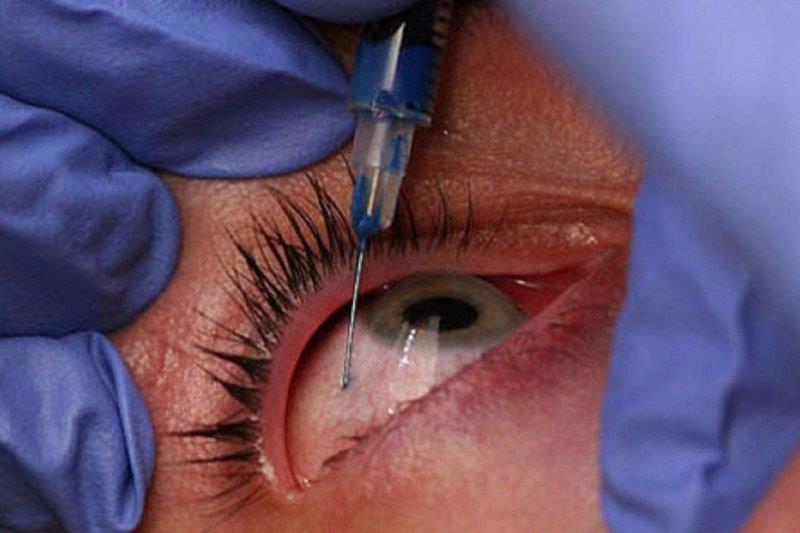
In March 2022, the breakthrough gene therapy treatment Luxturna was approved for public funding in Australia, two years after the UK did so and four years after it was fast-tracked to become the first FDA-approved gene therapy. Designed for people with vision loss caused by inherited retinal dystrophy from RPE65 gene mutations, including children with Leber’s congenital amaurosis (LCA), patients must have sufficient viable retinal cells to be eligible for treatment. However, the biggest barrier between New Zealand patients and Novartis’ sight-returning drug is its cost at nearly NZ$675,000 per eye, excluding surgical expenses.
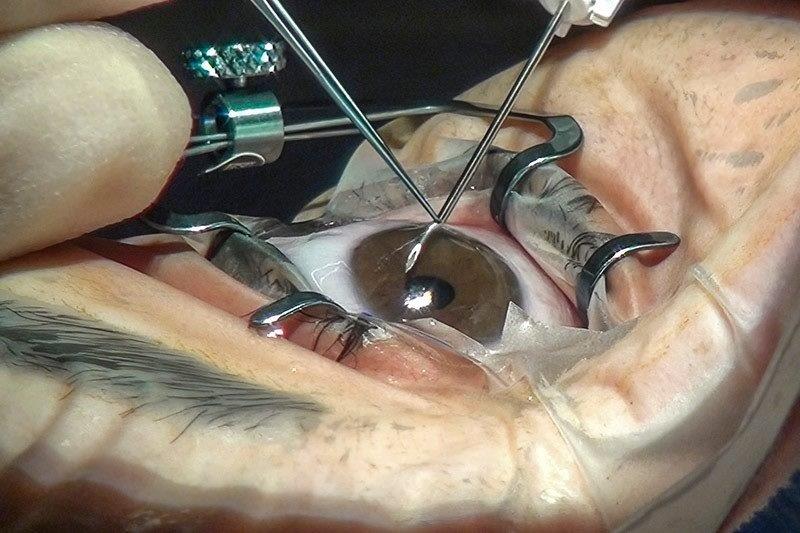
Leading the campaign to get Luxturna approved and funded in New Zealand is Auckland University Associate Professor Andrea Vincent, a consultant ophthalmologist specialising in genetic eye diseases at Auckland’s Retina Specialists and Greenlane Eye Clinic. Alongside Novartis submitting an application for Luxturna to the New Zealand Medicines and Medical Devices Safety Authority (Medsafe), establishing the appropriate pathways, skills and facilities for Luxturna treatment is vital in persuading the government’s drug funding agency Pharmac to fund it, she said. “In Auckland we are working to get accreditation for our surgeons; a new microscope with intraoperative OCT is on its way; we have all the appropriate investigations, including full-field stimulus testing, an electrodiagnostic test for which we have just upgraded at Retina Specialists; as well as working with Pharmac to get the appropriate standards.” These measures also will equip the country to administer other emerging gene-related therapies in advanced clinical trials, should they prove successful, she said.
There are three patients currently eligible for Luxturna treatment in New Zealand, said A/Prof Vincent, adding that although it’s expensive, there are multiple analyses showing the cost benefits and improvement in quality-adjusted life-years (QALY) for those treated. “I would encourage all ophthalmologists with patients with early onset severe retinal dystrophies to refer them for assessment and genotyping, so they do not miss out on a potential treatment.”
Having laid the foundations in New Zealand and following Australia’s decision, A/Prof Vincent said she remains optimistic for the drug making its way here. “Luxturna is a game changer and makes RPE65 early onset retinal dystrophy a treatable disease. To my mind, not treating it is negligence.”
For more, see https://www.nzoptics.co.nz/articles/archive/landmark-au-funding-for-luxturna-ird-treatment